Flexible and convenient dosing
For all VRAYLAR patients
Recommended dosing by indication
1.5 mg is the starting dose and a therapeutic dose for adjunctive therapy to antidepressants for MDD1
Recommended dosing in adults1

1.5 mg/day
Capsule image is not actual size.
- Day 1–Start at 1.5 mg/day
- Day 15–May increase dose to 3 mg/day
- Depending upon clinical response and tolerability, the dosage can be increased to 3 mg once daily at Day 151
- Following discontinuation of VRAYLAR, the decline in plasma concentrations of active drug and metabolites may not be immediately reflected in patients’ clinical symptoms1
- In clinical trials, dosage and titration at intervals of less than 14 days resulted in a higher incidence of adverse reactions. Maximum recommended dose is 3 mg once daily.1 (See adverse reactions from 8-week study in Section 6.1 of the full Prescribing Information)
1.5 mg is the starting dose and a therapeutic dose for bipolar I depression1
Bipolar I depression recommended dosing in adults1

1.5 mg/day
Capsule image is not actual size.
- Day 1–Start at 1.5 mg/day
- Day 15–May increase dose to 3 mg/day
Depending upon clinical response and tolerability, the dosage can be increased to 3 mg once daily at Day 15.
Maximum recommended dosage is 3 mg once daily.
Following discontinuation of VRAYLAR, the decline in plasma concentrations of active drug and metabolites may not be immediately reflected in patients’ clinical symptoms.1
Flexible and convenient dosing
Bipolar I acute manic or mixed episodes recommended dosing1
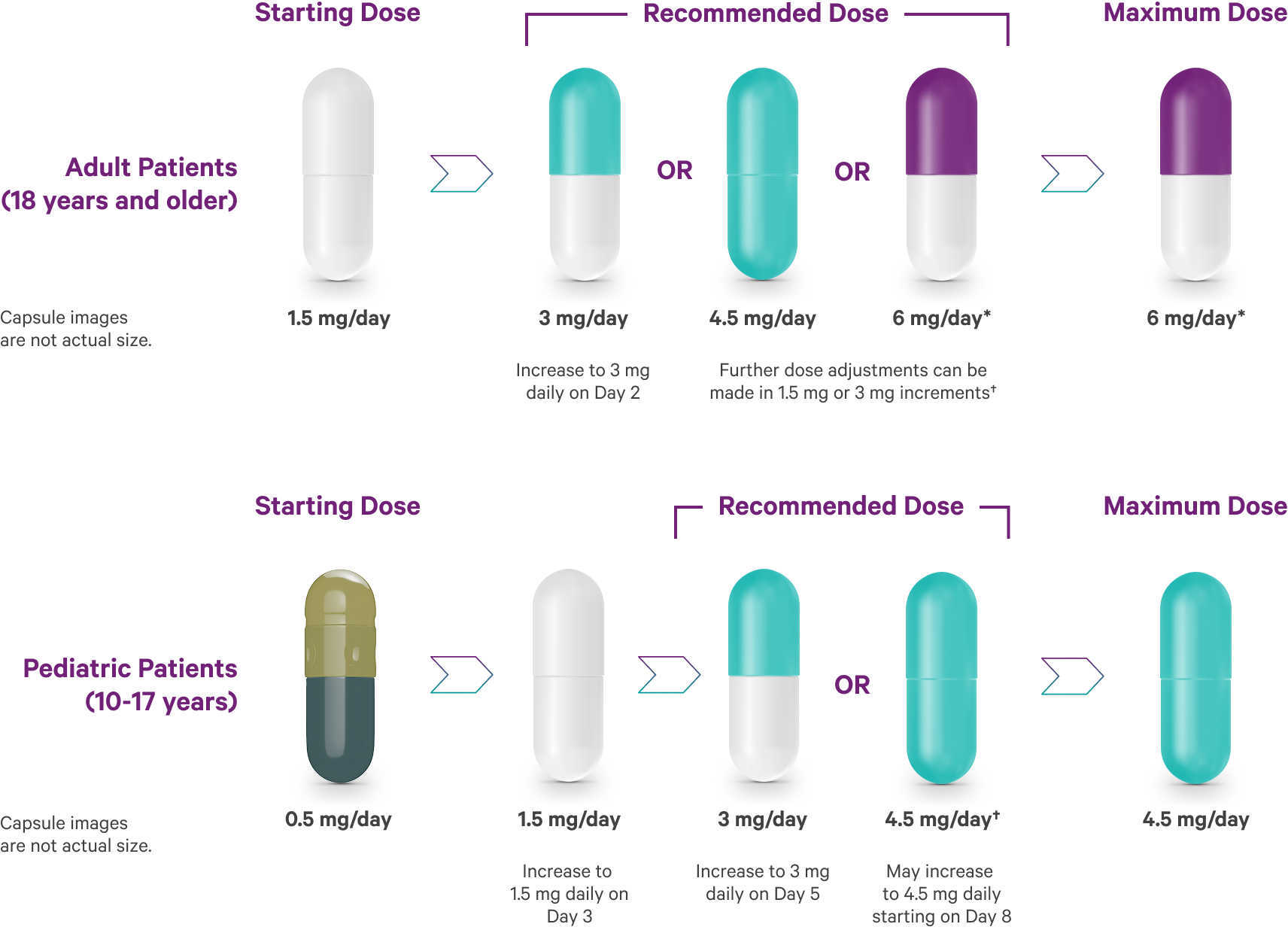
Following discontinuation of VRAYLAR, the decline in plasma concentrations of active drug and metabolites may not be immediately reflected in patients' clinical symptoms.1
*In short-term controlled studies in adults, doses above 6 mg/day do not confer increased effectiveness sufficient to outweigh dose-related adverse reactions.1
†Depending on clinical response and tolerability.
Flexible and convenient dosing
Schizophrenia recommended dosing1
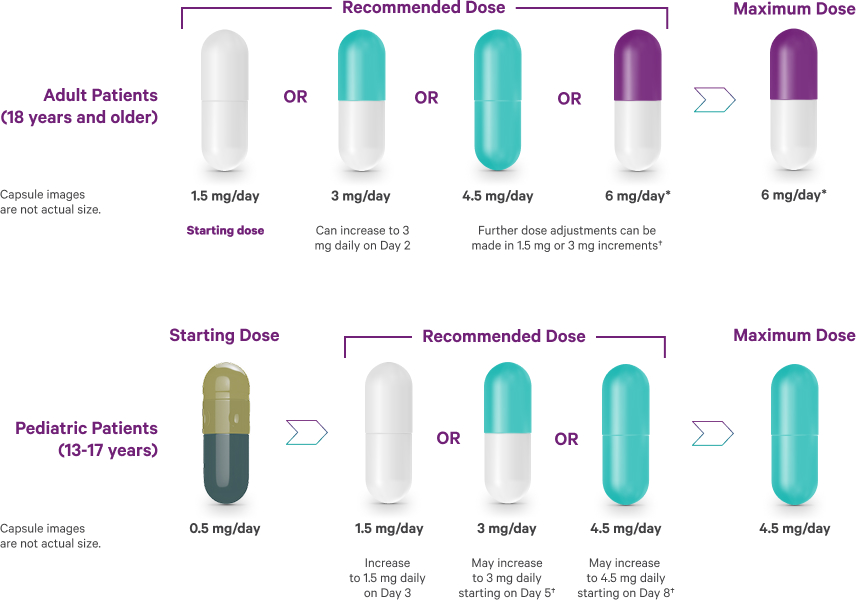
Following discontinuation of VRAYLAR, the decline in plasma concentrations of active drug and metabolites may not be immediately reflected in patients' clinical symptoms.1
*In short-term controlled studies in adults, doses above 6 mg/day do not confer increased effectiveness sufficient to outweigh dose-related adverse reactions.1
†Depending on clinical response and tolerability.
Dosing adjustments
No VRAYLAR dosage adjustment required based on1:
- VRAYLAR is not recommended in patients with severe hepatic (Child-Pugh score 10–15) or renal impairment (creatinine clearance <30 mL/min), as it has not been evaluated in these patient populations1
- VRAYLAR can be coadministered with a PPI. It does not affect VRAYLAR exposure at steady state1
PPI=proton pump inhibitor.
Drug interactions
Low doses are available for patients taking strong or moderate CYP3A4 inhibitors

0.5 mg

0.75 mg
Capsule images are not actual size.
VRAYLAR is extensively metabolized by CYP3A4 and to a lesser extent by CYP2D6.1
CYP3A4 is responsible for the formation and elimination of the major active metabolites of cariprazine.1
Dosage Modifications for CYP3A4 Inhibitors and Inducers
| Clinical impact | Intervention |
|---|---|
Strong CYP3A4 inhibitors |
Initiating strong CYP3A4 inhibitor while on a stable dose of VRAYLAR in adult and pediatric patients
Initiating VRAYLAR therapy while already on a strong CYP3A4 inhibitor
When a strong or moderate CYP3A4 inhibitor is withdrawn, VRAYLAR dosage may need to be increased*‡ |
Moderate CYP3A4 inhibitors |
Initiating moderate CYP3A4 inhibitor while on a stable dose of VRAYLAR in adult and pediatric patients
Initiating VRAYLAR while already on a moderate CYP3A4 inhibitor
When a strong or moderate CYP3A4 inhibitor is withdrawn, VRAYLAR dosage may need to be increased*‡ |
CYP3A4 inducers |
Concomitant use of VRAYLAR with a CYP3A4 inducer has not been evaluated and is not recommended |
*‡Depending upon clinical response and tolerability.
VRAYLAR does not require increased monitoring for serotonin reuptake inhibitor (SRI)-associated adverse events when used with an SRI1
Monitor patients for adverse reactions and treatment response for several weeks after starting VRAYLAR and after each dose change1
Select Examples of Strong and Moderate CYP3A4 Inhibitors
It’s important to talk to your patients about all the medications and products they are taking and to review the VRAYLAR Prescribing Information for more information on drug interactions.
Pharmacokinetics
~3-6 hours to peak plasma concentration1
after 1 dose of VRAYLAR 1.5 mg/day
~7-day half-life1
VRAYLAR plasma concentration decreased by ~50% in approximately 1 week‡∥
‡∥After multiple dose administration of VRAYLAR, mean cariprazine and DCAR concentrations reached steady state at around Week 1 to Week 2 and mean DDCAR concentrations approached steady state around Week 4 to Week 8 in a 12-week study. The half-lives based on time to reach steady state were 2 to 4 days for cariprazine, ~1 to 2 days for DCAR, and approximately 1 to 3 weeks for DDCAR. The time to reach steady state for DDCAR was variable across patients, with some not achieving steady state at the end of the 12-week treatment. After discontinuation of VRAYLAR, mean plasma concentrations of cariprazine and DCAR decreased by ~50% in ~1 day, and mean plasma concentration of DDCAR decreased by ~50% 1 week after the last dose. The decline in plasma concentrations of active drug and metabolites may not be immediately reflected in patients’ clinical symptoms.1
BP-I=bipolar I disorder; DCAR=desmethyl cariprazine; DDCAR=didesmethyl cariprazine; MDD=major depressive disorder.










